OUR TECHNOLOGY
Regensight is the world’s first Company introducing theranostics in vision care
Theranostics
(therapy guided by imaging diagnostics) is a combination of therapy and diagnostics; it is a revolutionary paradigm of personalized, predictive and precision medicine, which makes use of light activated image-guided drug therapy to achieve the best outcome in the treatment of visual disorders.

Regensight has developed a theranostic platform to treat demanding visual disorders in young people, such as myopia and keratoconus, and in the elderly, such as presbyopia.
Out platform enables surgeons in precisely tailoring the therapeutic UV-A light energy dose based on patterned riboflavin concentration in the cornea. We set a new, incision-free, standard in vision care.
THERANOSTICS is a precise, predictive therapy guided by real time assessment of treatment response.
The platform is the world’s first eye-care device using the IoMT (Internet of Medical Things) technology providing remote control for predictive maintenance, improving clinical research data collection and product improvement.
The Platform holds “fleet’s learning” ability, thus making Regensight as the world’s first biomedical manufacturer of an adaptive surgical platform.
THERANOSTICS is an intelligent surgical therapy, which is driven by real time image analysis of the eye under treatment.
OUR PROJECTS
Regensight has developed a theranostic platform incorporating a sophisticated UV-A medical device and a controlled delivery device for personalized delivery of proprietary 0.22% riboflavin ophthalmic formulation into the cornea
We are working to offer eye surgeons a new, incision-free and risk-free, approach leading to precise and effective correction of the most frequent visual disorders in young and old people.
| Idea | Pre - Clinical | Prototyping | Industralization | Clinical |
|---|
| Approved |
|---|
| C4V CHROMO4VIS |
| - |
| - |
Our Short-Term Plan is focused on introducing theranostics for developing new treatment options for improving vision in keratoconus patients.
Our Mid-Term Plan is focused on developing revolutionary treatment options for people with low-grade myopia and presbyopia.
Our approved products
Currently, Regensight manufactures three CE marked medical devices for the indication of use of treating keratoconus and corneal disorders with theranostic-guided corneal cross-linking procedures. Learn More
Clinical Trials
Our aim is to validate theranostics for the correction of visual disorders
Unique in theranostic-guided correction of visual disorders, we aim to validate our new paradigm based on proprietary technology to treat a wide range of visual disorders.
If you wish to participate or have any question about our clinical trials, please email us at clinicaltrials@regensight.com
Currently, there are no ongoing randomized clinical trials.
VISUAL DISORDER
What are visual disorders?
Visual disorders, or refractive errors, are the most frequent causes of visual impairment worldwide. They can be caused by optical distortions (aberrations) of the eye or by a change in the axial length of the eye or by a decreased function of the intraocular muscles.The main refractive disorders causing visual impairment are:
Keratoconus
is the primary cause of corneal transplantation in young people worldwide.
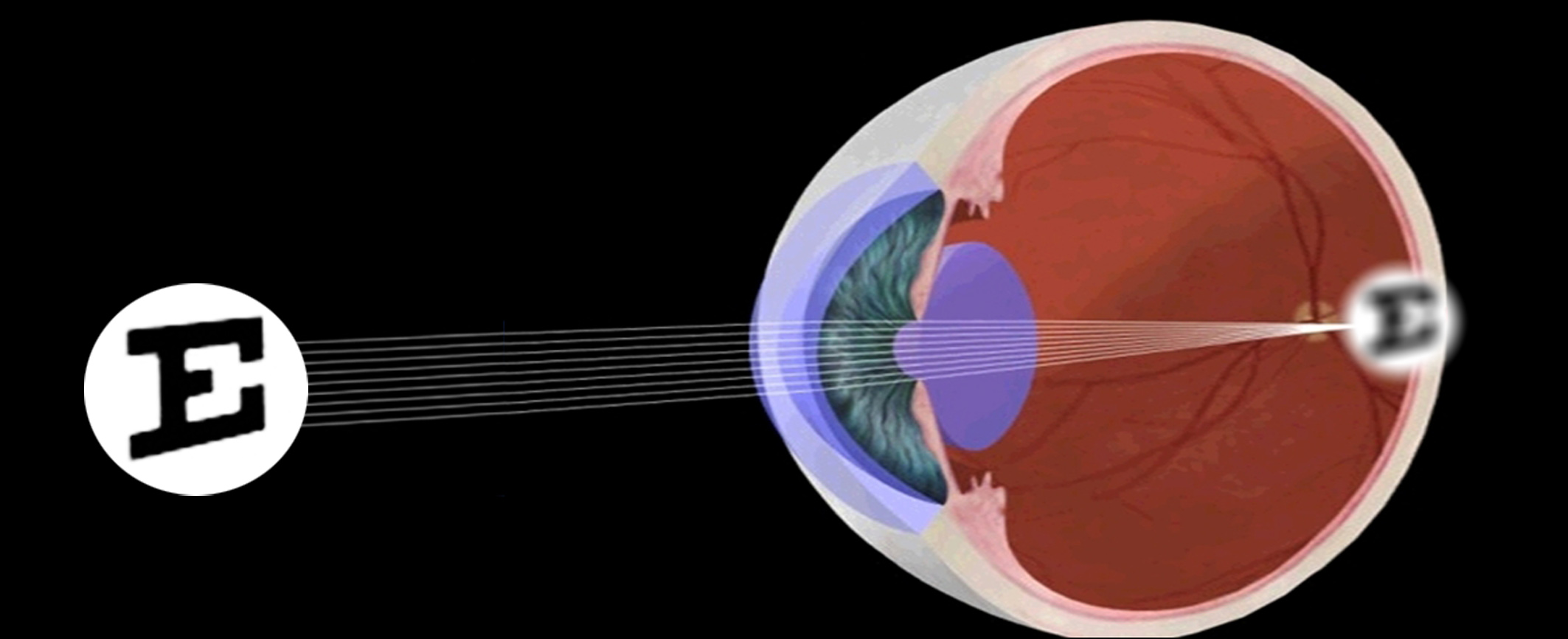
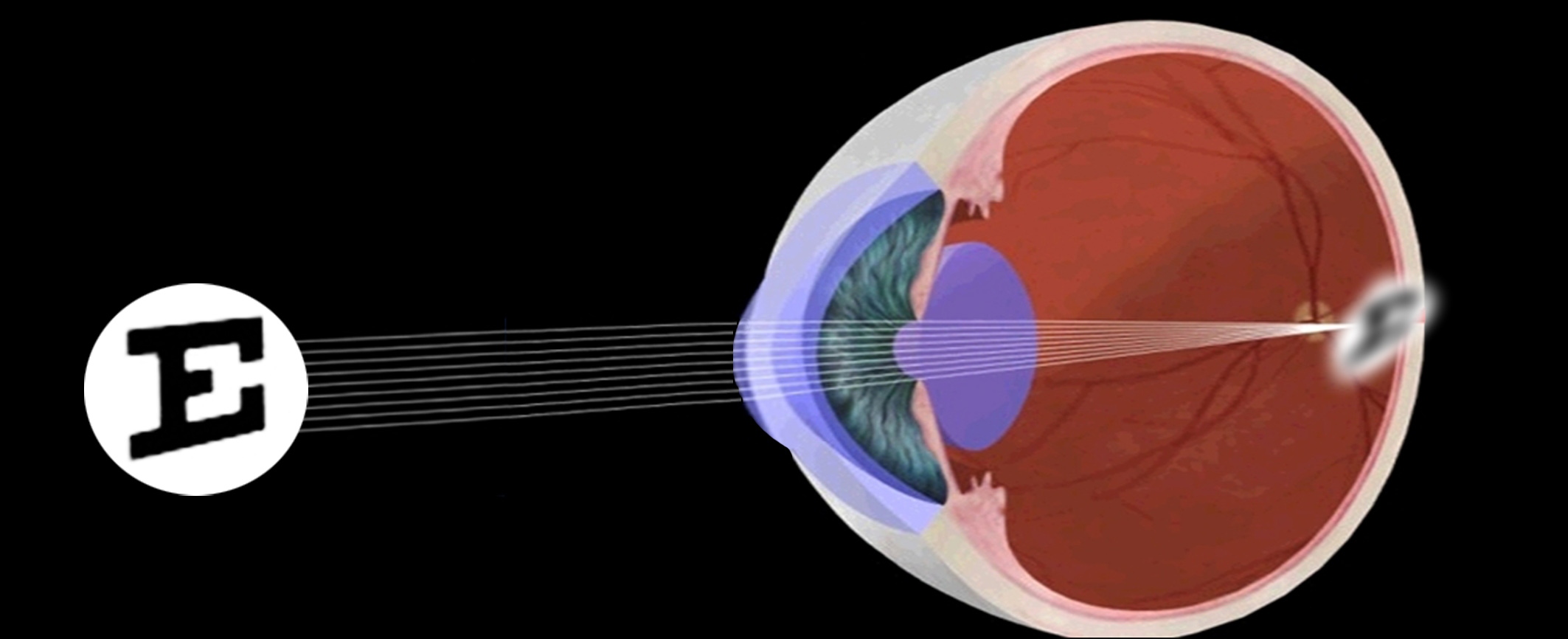
Myopia
caused by longer eye axial length. It is the primary cause of visual impairment in young people.
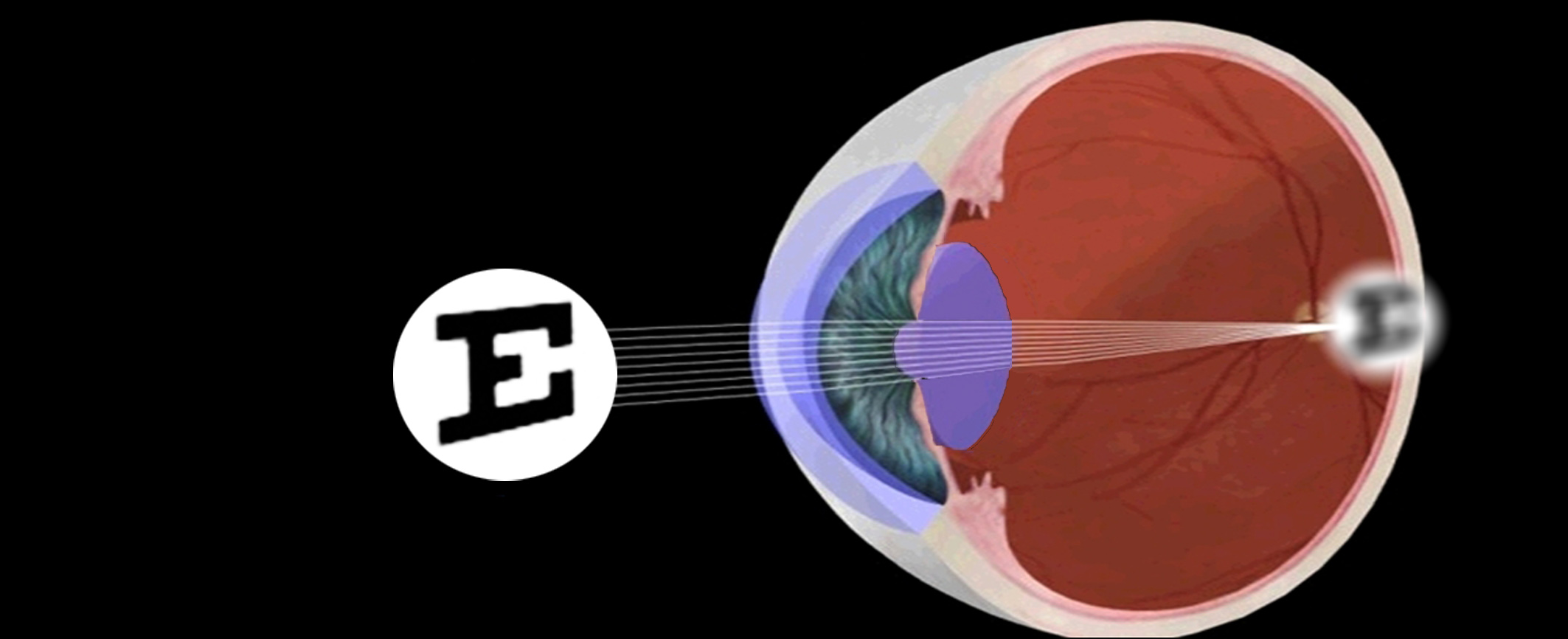
Presbyopia
caused by a decrease in accommodation. It is the primary cause of visual impairment in the elderly.
Limits of current medical technologies
Current surgical treatments of myopia, such as laser correction, although effective, have known problems like pain and the risk of infection and corneal scarring. In addition, laser correction is contraindicated in at least one third of myopes, because the cornea is thin or has keratoconus.
Until today there is no effective and safe surgical solution for the most frequent visual disorder in the elderly, such as presbyopia.
Corneal cross-linking with riboflavin and UV-A light could be effective in halting keratoconus progression only if the corneal epithelium is removed; however, the epithelium removal has 15% rate of complications and the procedure does not improve visual acuity, which remains low due to the corneal distortion.
Current limits of riboflavin/UV-A corneal cross-linking
Efficacy of corneal cross-linking, intended as stabilization of disease progression, is highly variable (from 10% to 90%) using the standard protocol (i.e., the cornea is de-epithelialized). This variability forces eye surgeons to prefer the standard protocol, which, however, has high complication rates (incidence: 15%).
UV-A medical devices without theranostics have several technical limits:
- Do not provide information on the concentration and UV-A photo-activation of riboflavin into the cornea, which is the main parameter determining treatment efficacy;
- Do not assess treatment efficacy, causing a delay in the assessment of disease progression and inadequate patient care;
- Do not provide a full range of treatment operations to the surgeon;
- Do not maintain maximum device realiability during operation.
Publications
The pre-clinical and clinical evidences on efficacy and safety of the Regensight platform for treatment of keratoconus are regularly published in high impact factor scientific journals:
J Biophotonics 2025b;18(2):e202400462.
Ophthalmology 2024: S0161-6420(24)00367-1d.
J Biophotonics 2024; 17(7):e202400068
Graefe’s Arch Clin Exp Ophthalmol 2024; 262: 2569-2577
Lombardo G, Bernava GM, Serrao S, M, Roszkowska AM, Lombardo M. Predicting corneal cross-linking efficacy with real-time assessment of corneal riboflavin concentration.
J Cataract Refract Surg 2023; 49(6): 635-641.
Roszkowska AM et al. A randomized clinical trial assessing theranostic-guided corneal cross-linking for treating keratoconus: the ARGO Protocol.
Int Ophthalmology 2022 Dec; E-pub ahead of print.
Lombardo G, Bernava GM, Serrao S, Lombardo M. Theranostic-guided corneal cross linking: pre-clinical evidence on a new treatment paradigm for keratoconus.
J Biophotonics 2022; 15(12):e202200218.
Lombardo G et al. Non-invasive optical method for real-time assessment of intracorneal riboflavin concentration and efficacy of corneal cross-linking.
J Biophotonics 2018;11(7):e201800028.
Lombardo M, Lombardo G. Non-invasive and real time assessment of riboflavin consumption in standard and accelerated corneal cross-linking.
J Cataract Refract Surg 2019; 45(1):80-86.
Lombardo G et al. Comparison between standard and transepithelial corneal cross-linking using a theranostic UV-A device.
Graefe’s Arch Clin Exp Ophthalmol 2020; 258(4): 829-834.
Lombardo M, Lombardo G.
Grand Round Stanford
Lombardo M, Lombardo G.
Grand Round UCL

